China’s Sinopharm Vaccine Put on WHO Emergency Use List

China’s Sinopharm Covid-19 vaccine has won World Health Organization (WHO) approval for emergency use, making it the sixth shot to receive the body’s green light and paving the way for its broader rollout worldwide.
The WHO’s Friday nod represents its approval of the vaccine’s safety, efficacy and quality, according to its statement.
“The addition of this vaccine has the potential to rapidly accelerate COVID-19 vaccine access,” Mariângela Simão, WHO assistant-director general for access to health products, said in the statement.
The WHO authorization makes the Chinese vaccine eligible for use in Covax, a global program co-led by the organization and other health groups to guarantee that every country has fair access to Covid-19 vaccines. The initiative is believed to particularly benefit poor countries suffering vaccine shortages. The previous five vaccines that have won WHO approval for emergency use are the Pfizer/BioNTech, Janssen and Moderna shots, as well as two versions of the AstraZeneca/Oxford vaccine.
In recent weeks, multiple countries have turned to China for vaccines. On the one hand, the escalating Covid-19 crisis in India, a major vaccine exporter, has ramped up domestic demand and squeezed its supply to the rest of the world; on the other, the U.S., a major vaccine producer, has been criticized for its restrictions on exports of vaccine raw materials.
The WHO’s decision also means that people inoculated with the Sinopharm vaccine are likely to be able to travel more freely. On Monday, the EU proposed that its member states ease restrictions on inbound travel for people vaccinated with shots given WHO approval for emergency use.
Read more
Charts of the Day: Covid Vaccines By the Number
So far, China has given its nod for five Covid-19 vaccines for general or emergency use, with the Sinopharm shot being the first approved for use on the general public. The approval came in December, five months after the vaccine was granted (link in Chinese) the green light for domestic emergency use.
The vaccine is produced by a Beijing subsidiary of state-owned pharma giant China National Pharmaceutical Group Co. Ltd., better known as Sinopharm.
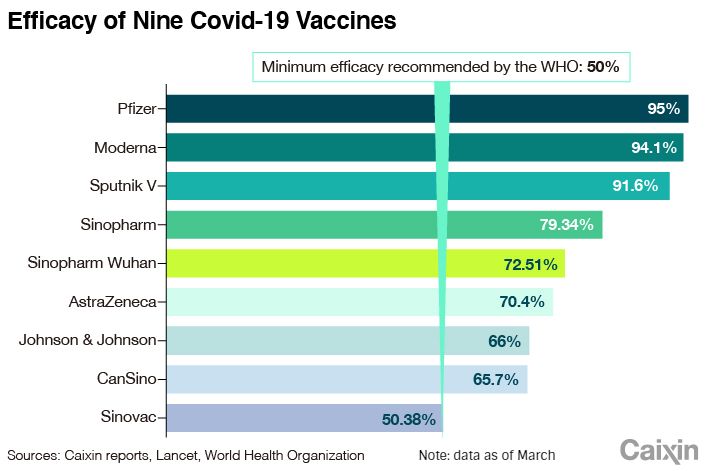
The two-dose vaccine’s efficacy for preventing symptomatic and hospitalized disease was estimated to be 79% for all age groups combined, the WHO said. It recommends the vaccine for adults 18 years and older, while efficacy for those over 60 could not be estimated due to lack of clinical trial data, according to the statement.
The Sinopharm vaccine is the first shot granted the WHO approval that will carry a vaccine vial monitor, “a small sticker on the vaccine vials that change color as the vaccine is exposed to heat, letting health workers know whether the vaccine can be safely used,” the statement said.
So far, the Sinopharm vaccine and another Covid-19 shot developed by the company’s Wuhan subsidiary have been sold or donated to more than 60 countries and regions (link in Chinese), including Argentina, Myanmar and the United Arab Emirates, according to a statement it released on Friday.
 |
The WHO will soon decide whether to also approve another Chinese vaccine for emergency use, which was developed by Sinovac Biotech Ltd. A working group on Covid-19 vaccines at the WHO has conducted its review, saying that it is “very confident” that the vaccine is efficacious in preventing Covid-19 in adults aged 18 to 59.
It has been about a year and a half since the first known case of Covid-19 was detected in the central Chinese city of Wuhan. So far, the novel coronavirus has infected about 157 million people and claimed more than 3.2 million lives worldwide, according to data compiled by Johns Hopkins University.
The U.S. has recorded the most cases, totaling more than 32 million, followed by India, the world’s second-most populous nation, which has reported about 22 million cases.
Contact reporter Lin Jinbing (jinbinglin@caixin.com) and editor Joshua Dummer (joshuadummer@caixin.com)
Download our app to receive breaking news alerts and read the news on the go.
Follow the Chinese markets in real time with Caixin Global’s new stock database.

- MOST POPULAR






